Botulism is a bacterial food-borne infectious disease, not vector-borne or climate-related, so it fits squarely under Infectious Diseases (and broadly under Public Health). Don’t tick Mosquito-Borne / Vector-Borne / Women Health for this one.
If you’re a parent in the U.S. and you’ve seen “botulism” trending, you’re not imagining it—and you’re not overreacting by being worried.
In late 2025, U.S. health authorities confirmed a multistate outbreak of infant botulism linked to ByHeart Whole Nutrition infant formula.(CDC) On November 11, 2025, ByHeart, in coordination with the FDA, recalled all lots of its powdered infant formula products nationwide, after earlier recalling just two batches.(CDC)
As of November 14, 2025, the CDC and FDA report 23 infants with suspected or confirmed infant botulism across 13 U.S. states. All affected babies have been hospitalized, and no deaths have been reported so far.(CDC)
This is not a routine recall over minor labeling issues. Infant botulism is rare but medically serious: it can cause muscle weakness, breathing problems, and weeks to months of hospital care if not treated quickly.(CDC)
This article will walk you through, in plain language:
- What is actually happening in this outbreak
- Which ByHeart products are affected
- What risk this poses to your baby
- Symptoms you must watch for
- The exact steps you should take today if your child ever used ByHeart formula
The goal isn’t to scare you—it’s to give you clear, practical instructions so you can act fast and intelligently instead of scrolling through panic posts and half-truths.
Quick Facts – The Outbreak at a Glance
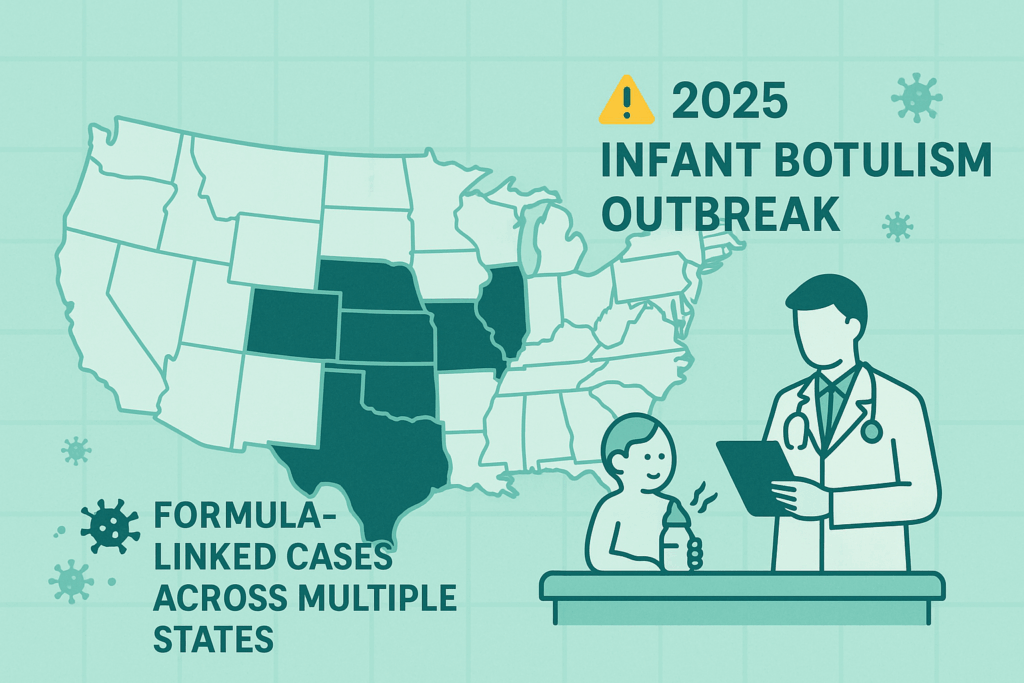
If you just need the essentials first, here’s the current snapshot.
- What’s happening?
A multistate outbreak of infant botulism has been linked to ByHeart Whole Nutrition infant formula. The CDC, FDA, and multiple state health departments are actively investigating.(CDC) - How many babies are affected?
- 23 infants with suspected or confirmed infant botulism
- All 23 hospitalized
- No deaths reported as of November 14, 2025(CDC)
- Where are the cases?
- 13 U.S. states have reported affected infants (CDC’s outbreak map confirms this count).(CDC)
- New individual state alerts (e.g., Washington, New York, California, Vermont, Kansas, Kentucky) confirm local cases linked to the same recall.(King County)
- Which product is involved?
- ByHeart Whole Nutrition Infant Formula
- All canned products (24 oz) and Anywhere Pack™ single-serve sticks
- All lots are under recall—this is not limited to just a couple of batch numbers.(CDC)
- What has ByHeart done?
- Nov 8, 2025: voluntary recall of two lots after early investigation signals.(California Department of Public Health)
- Nov 11, 2025: recall expanded to all ByHeart Whole Nutrition infant formula products nationwide, following FDA’s broader investigation.(CDC)
- How rare is this?
- Infant botulism is very rare, even now. Since August 2025, FDA has been notified of dozens of infant botulism cases nationwide, of which a subset had exposure to ByHeart formula.(U.S. Food and Drug Administration)
- But because botulism can cause life-threatening paralysis and breathing failure, regulators treat even a small cluster extremely seriously.(CDC)
- Has any unopened formula tested positive?
- So far, preliminary testing found possible Clostridium botulinum in one opened can from a sick infant’s household.
- No unopened cans have tested positive yet, which is part of why the company is publicly disputing a direct causal link—however, CDC/FDA still see a strong epidemiologic association and have not backed off the recall.(California Department of Public Health)
- Is there a nationwide formula shortage because of this?
- No. ByHeart represents well under 1% of U.S. infant formula sales, and health authorities do not expect this recall alone to trigger a national shortage.(U.S. Food and Drug Administration)
- What should parents do right now?
- Stop using any ByHeart formula immediately.
- Do not throw it away yet – keep the product and take photos of the lot information in case public health needs it.(CDC)
- Watch your baby closely for symptoms (constipation, poor feeding, weak cry, floppy head, breathing changes) for several weeks after last use.
- Seek urgent medical care if any warning signs appear.
What Is Infant Botulism – and Why It’s So Serious?
Before we talk more about the recall, you need to understand what infant botulism actually is. Otherwise the headlines just sound like generic panic.
At its core, infant botulism is a toxin problem, not a “normal infection” problem.
The simple version
- A bacterium called Clostridium botulinum produces one of the strongest nerve toxins known.
- In older children and adults, we usually see botulism when pre-formed toxin is swallowed (for example from badly canned food).
- In infants, the story is different: they can swallow spores (tiny dormant forms of the bacteria) from food or the environment. Those spores can then germinate inside the baby’s gut, start growing, and produce toxin there. (CDC)
That toxin blocks the signals between nerves and muscles. When enough of those signals are blocked, muscles simply don’t work properly.
For a baby, that shows up as:
- less movement,
- weaker sucking and crying,
- loss of head control,
- and, in severe cases, trouble breathing. (Washington State Department of Health)
So when health departments stress this outbreak, it’s not because lots of babies are dying (so far, no deaths have been reported); it’s because even a single untreated case can end up on a ventilator.
How babies usually get infant botulism (in “normal” years)
Historically, infant botulism cases have almost never been linked to commercial infant formula. The usual pattern is: (CDC)
- The infant is exposed to dust or soil containing C. botulinum spores (for example in certain rural or construction areas), and/or
- The baby is given honey before 12 months of age (honey is a well-known source of spores).
Most cases are sporadic, meaning one baby in one family, not clusters across many states. Public health teams investigate, maybe find some environmental sources, the child is treated with antitoxin, and the story ends there.
That’s why this situation is different.
Why this outbreak stands out
Three things make the current situation unusual:
- Clustering in time
Health agencies saw a noticeable spike in infant botulism reports starting in August 2025, instead of the usual low, stable background level. (U.S. Food and Drug Administration) - A common product link
When CDC and state teams went through feeding histories, they kept seeing ByHeart Whole Nutrition Infant Formula show up in a disproportionate number of cases compared with its tiny market share. (U.S. Food and Drug Administration) - Lab signal from an opened can
In at least one household, an opened can of ByHeart formula used by a sick infant gave preliminary evidence of C. botulinum contamination when tested. (foodpoisonjournal.com)
Put together, that’s enough for CDC and FDA to treat this as an outbreak linked to infant formula, even though definitive “smoking gun” lab confirmation from unopened cans is still being worked through.
For parents, the practical translation is simple:
Infant botulism remains rare, but when multiple cases line up in time, across states, with the same product, regulators don’t gamble—they assume the worst-case route and move fast.
How ByHeart Infant Formula Got Linked to the 2025 Outbreak
The recall didn’t come out of nowhere. There’s a clear timeline of how ByHeart went from “one of many niche formula brands” to the center of a national botulism investigation.
Step 1 – A spike in rare cases sets off alarms
From August 2025 onward, state health departments and the California Department of Public Health’s Infant Botulism Treatment and Prevention Program noticed they were seeing more infant botulism cases than usual. (CDC)
Infant botulism is rare enough that even a modest spike is suspicious. California flagged the pattern to CDC, which started pulling data from multiple states and the FDA.
At this stage, nobody is blaming a specific product yet. The question is simply:
“Why are we suddenly getting so many infant botulism reports?”
Step 2 – Case interviews reveal a common thread
Whenever there’s an unusual cluster, epidemiologists go back to basics: interviews.
For each infant, they look at:
- feeding history (breastfeeding, formula brand, any changes),
- home environment (dust, soil exposures, renovations),
- any supplements, honey, herbal products, etc.
When CDC and FDA combined the case investigations, they noticed that a chunk of the affected infants had been given ByHeart Whole Nutrition Infant Formula, often as a main or regular formula, even though ByHeart holds well under 1% of the U.S. formula market. (U.S. Food and Drug Administration)
That imbalance is exactly the kind of signal outbreak investigators look for.
Step 3 – Lab work adds weight to the suspicion
While interviews were happening, labs were also testing stool samples from infants and, in some homes, remaining formula.
- C. botulinum was confirmed in infants’ stool in multiple states, consistent with infant botulism. (CDC)
- Critically, one opened can of ByHeart formula from the home of an affected infant showed preliminary evidence of C. botulinum contamination when tested, according to several state and media reports. (foodpoisonjournal.com)
FDA is cautious by design: they have not publicly declared that unopened formula on shelves is positive for toxin-producing C. botulinum. But epidemiologic patterns + that kind of lab signal are enough to justify strong action.
Step 4 – The first limited recall (two batches)
On November 7, 2025, FDA informed ByHeart that among roughly 80+ infant botulism cases reported nationwide since August, 13 babies had received ByHeart at some point, and that one opened can showed possible contamination. (U.S. Food and Drug Administration)
Within a day, ByHeart initiated a voluntary recall of two specific batches of their Whole Nutrition Infant Formula. (U.S. Food and Drug Administration)
That was the “narrow” recall phase—focused on the most clearly implicated lots.
Step 5 – The recall expands to all ByHeart formula
Over the following days, more case data came in from other states. By November 10–11, FDA’s outbreak page stated that 15 infants with suspected or confirmed infant botulism had documented exposure to ByHeart formula across 12 states, all hospitalized. (U.S. Food and Drug Administration)
At the same time, state advisories described 23 infants in 13 states with suspected/confirmed infant botulism in the broader outbreak, reinforcing the sense that this was not a local fluke. (Cabell-Huntington Health Department)
Because illnesses were tied to multiple different lot numbers, not just the first two, the FDA and CDC pushed for a broader action.
On November 11, 2025, ByHeart expanded its voluntary recall to all unexpired cans and Anywhere Pack™ sticks of ByHeart Whole Nutrition Infant Formula across the U.S. (U.S. Food and Drug Administration)
That’s the recall you’re now seeing in national headlines.
Step 6 – The company’s public stance vs. regulators’ stance
ByHeart’s public messaging has tried to make two points at once:
- They emphasize that no unopened can has yet tested positive for C. botulinum, and that FDA has not proven a direct causal link.
- They also say they expanded the recall “out of an abundance of caution” while fully cooperating with the investigation. (ByHeart)
Food safety experts, on the other hand, have criticized some of the company’s tone and framing, arguing that it risks confusing parents when the priority should be extremely clear: do not use the product until the investigation concludes. (STAT)
From a parent’s perspective, the distinctions don’t change the practical reality:
CDC and FDA have both publicly tied the outbreak to ByHeart formula, and every unexpired ByHeart Whole Nutrition Infant Formula product is under recall. That’s the level of risk regulators are working with.
Step 7 – Where things stand right now
As of mid-November 2025:
- The recall remains nationwide and comprehensive for ByHeart Whole Nutrition Infant Formula (cans and Anywhere Packs). (Piedmont Pediatrics)
- The outbreak investigation is still active, with CDC, FDA, California’s Infant Botulism Program, and multiple state health departments coordinating lab work and case tracking. (CDC)
- More detailed lab results from product testing may still emerge, but regulators are already acting as if any ByHeart formula is potentially unsafe for infants.
For you, the action plan is straightforward: treat every ByHeart can or stick in your home as recalled, stop using it, and move on to a different formula brand under your pediatrician’s guidance.
Which ByHeart Products Are Actually Recalled?
Let’s clear this up first, because many parents are still asking, “Is my can part of the recall or not?”
Right now, this isn’t a “few bad batches” type of recall anymore. It’s everything under the ByHeart Whole Nutrition infant formula line.
According to CDC and FDA: (U.S. Food and Drug Administration)
All ByHeart Whole Nutrition Infant Formula products are recalled.
That means:
- All lot numbers
- All can sizes
- All single-serve packets (Anywhere Pack™ sticks)
- All unexpired product, nationwide
What exactly is included?
Based on the latest outbreak and recall notices: (U.S. Food and Drug Administration)
- ByHeart Whole Nutrition Infant Formula – 24 oz powdered cans
- ByHeart Whole Nutrition Infant Formula – single-serve “Anywhere Pack™” sticks
These products were sold:
- Directly from ByHeart’s website
- Major online marketplaces (e.g. Amazon-type platforms)
- Large brick-and-mortar retailers (national chains and supermarkets)
If it’s ByHeart Whole Nutrition powdered infant formula and it’s not expired yet, treat it as recalled, full stop.
How to check your can (and what to do with it)
- Find the product details
- Look at the front label to confirm the brand and product name: “ByHeart Whole Nutrition Infant Formula”
- Flip the can or box and find the lot number and “use by” date (usually printed on the bottom or side).
- Assume recall if it’s ByHeart Whole Nutrition
- Don’t waste time trying to match lot codes against some partial list from early November.
- CDC and FDA have explicitly said: all lots, all sizes, all packets of ByHeart Whole Nutrition infant formula are now covered. (CDC)
- Do not keep feeding it “until it’s finished”
- There is no “safe” leftover portion from a recalled container.
- Even if your baby seems fine today, you stop using it now.
- Don’t toss it straight in the trash yet
- Health advisories repeatedly tell parents:
- Take clear photos of the front of the can and the lot/best-by area. (Tacoma-Pierce County Health Department)
- Write “DO NOT USE – RECALLED” on the lid or box.
- Store the product somewhere out of reach of children and pets.
- Reason: public-health teams may need details or samples, and some retailers or the company may offer refunds or replacements based on proof of purchase.
- Health advisories repeatedly tell parents:
- Check official recall information (not random social posts)
- FDA outbreak page – “Outbreak Investigation of Infant Botulism: Infant Formula (November 2025)” (U.S. Food and Drug Administration)
- CDC outbreak page – “Infant Botulism Outbreak Linked to Infant Formula” (CDC)
- Any state health advisory where you live (many states have posted their own ByHeart alerts). (Health Vermont)
Bottom line: if the label says ByHeart Whole Nutrition Infant Formula and it’s not expired, treat the entire product as recalled and unusable for your baby.
What Is the Real Risk for My Baby?
Let’s be blunt, because this is what you actually care about:
“My baby drank ByHeart formula. How scared should I be right now?”
You shouldn’t panic, but you also shouldn’t shrug it off. You need a realistic picture of the risk so you can act like a responsible adult, not a frightened zombie scrolling TikTok.
How big is the outbreak?
As of the latest FDA and CDC update (mid-November 2025): (U.S. Food and Drug Administration)
- 23 infants with suspected or confirmed infant botulism
- All had confirmed exposure to ByHeart Whole Nutrition infant formula
- Cases span 13 U.S. states
- All 23 infants were hospitalized
- No deaths reported so far
Most cases are in babies roughly 2 weeks to 6 months old, which is exactly the age window when they’re most vulnerable. (AP News)
Absolute risk vs. relative risk
This is where people get confused, because two things are true at the same time:
- Absolute risk is still low.
- There are millions of infants in the U.S., and 23 cases is still a small number in that context.
- Even among all babies who ever used ByHeart, only a minority have gotten sick.
- Relative risk for ByHeart-exposed infants is high enough to take very seriously.
- ByHeart makes <1% of formula sold in the U.S., but it shows up in a much bigger fraction of recent infant botulism cases. (U.S. Food and Drug Administration)
- That over-representation is why CDC and FDA didn’t hesitate to recommend a full-line recall.
So no, this is not “every ByHeart baby is doomed”. But if your baby had ByHeart, they are in a clearly higher-risk group compared with babies who never touched it. You treat that seriously.
How long after exposure can symptoms appear?
This is another key point many headlines skip.
CDC and multiple state health departments note: (U.S. Food and Drug Administration)
- Infant botulism symptoms can take days to several weeks to show up after swallowing the spores.
- FDA’s outbreak curve shows illness onset between August 9 and November 11, 2025, for the currently known cases. (U.S. Food and Drug Administration)
So if your baby finished ByHeart last week and is fine today, you are not completely “out of the woods” yet. You need to keep an eye on them for at least a few weeks after the last exposure.
Which babies should be most on alert?
You should be especially cautious if your baby:
- Is younger than 12 months (infant botulism is mainly a problem in this age group).
- Used ByHeart regularly as a main formula between August and now.
- Has recently shown any early warning signs:
- new or worsening constipation,
- poor feeding,
- unusually weak or soft cry,
- seems a bit floppy or struggles to hold their head,
- looks less expressive in the face than usual. (Piedmont Pediatrics)
If all three are true (under 1 year + regular ByHeart use + any of those symptoms), you treat this as urgent and call your pediatrician or go to the ER.
What if my baby seems totally fine?
If your baby had ByHeart but currently looks well:
- Stop the formula immediately (already covered above).
- Switch to a different formula with your pediatrician’s guidance.
- Watch closely for at least a month after the last ByHeart feed.
- Trust your gut: if you think your baby’s behaviour, feeding, or muscle tone is “not right”, don’t sit on it. Get them seen.
Remember: all 23 known affected infants have been hospitalized and treated, and none have died so far. (U.S. Food and Drug Administration) Early recognition and treatment with BabyBIG (the antitoxin) dramatically improves outcomes.
Hard truth you need to accept
If your baby used ByHeart:
- You can’t change that fact now.
- You can do three things that actually matter:
- Never feed a recalled product again.
- Monitor your child with a clear list of symptoms in mind instead of vague anxiety.
- Seek care fast if anything feels off.
That’s how you convert “headline panic” into real risk management for your own child.
Infant Botulism Symptoms Parents Must Know (In Plain Language)
Now we get to the part that actually changes outcomes: what you watch for in your baby.
Forget the scary word “botulism” for a minute. Think in terms of how your baby behaves and moves. That’s where the toxin shows itself.
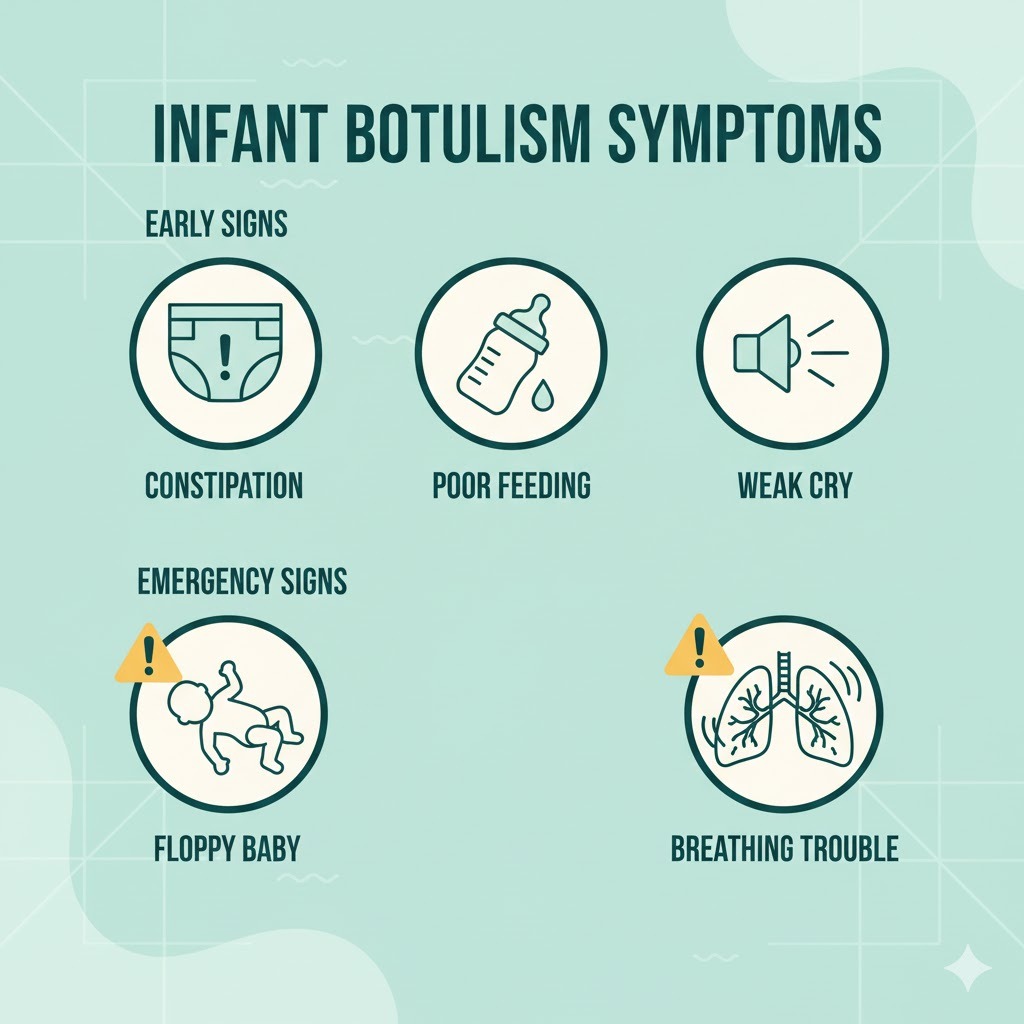
Early warning signs (the “something is off” stage)
These are the subtle changes that often appear first. If your baby has used ByHeart recently and you see any of these, you take them seriously:
- New or persistent constipation
Not “one slightly hard stool.” We’re talking about:- Fewer stools than usual for your baby plus
- Straining, or a clear change from their normal pattern.
- Poor feeding
- Baby latches but sucks weakly, gets tired quickly, or keeps falling asleep on the bottle or breast.
- You notice they’re taking less milk than usual or taking much longer to finish a feed.
- Weak or different-sounding cry
- Cry becomes softer, hoarse, or “thin”, compared with your baby’s normal loud cry.
- You feel like “this doesn’t sound like my baby”.
- Floppy head or reduced head control
- A baby who previously could hold their head up now struggles.
- Head feels heavier or flops back more when you pick them up.
- Less movement and expression
- Arms and legs move less.
- Baby looks “tired” or “blank”, smiles less, and facial muscles look weaker.
Any one of these in isolation can have other explanations, but in a baby who used ByHeart recently, you don’t ignore them.
Red-flag emergency symptoms (don’t sit at home with these)
If you see any of the following, you stop reading the internet and go to the ER or call emergency services:
- Noticeably floppy baby (“rag doll” feel)
- Limbs feel weak, the baby can’t fight against gravity the way they usually do.
- When you lift them under the arms, they slip through your hands more than usual.
- Difficulty swallowing or controlling saliva
- Milk dribbling out more than usual.
- Choking or coughing repeatedly during feeds.
- Lots of drooling with a sense they’re not managing it well.
- Breathing changes
- Breathing looks shallow, slow, or irregular.
- The skin around ribs or neck pulls in when they breathe (retractions).
- Any episode where baby looks blue around lips or face.
- Unusual sleepiness or unresponsiveness
- Hard to wake, not reacting to you the way they normally do.
- Eyes half-open, floppy, or not tracking you properly.
These signs mean the toxin may be affecting the muscles that control breathing and swallowing. That’s not something you “monitor at home”.
How doctors think about these symptoms
Clinically, doctors don’t wait for some perfect “botulism test” to come back. They look at a few key things together:
- Age (under 1 year, especially under 6 months).
- History of ByHeart formula use or other potential exposures.
- A pattern of constipation → weakness → feeding problems → breathing risk.
If that picture fits, they treat it as suspected infant botulism and start the right process (including arranging BabyBIG, the antitoxin) instead of wasting time.
Your job is not to diagnose. Your job is to notice early changes and get the baby in front of someone who can.
Step-by-Step: What To Do If You Have Used ByHeart Formula

This is the practical checklist. Don’t overcomplicate it.
There are two big scenarios:
- You still have ByHeart at home (opened or unopened).
- You already used it all, but your baby drank it in the last few weeks.
The steps are almost the same.
Step 1 – Stop using ByHeart immediately
This is non-negotiable.
- Do not “finish the can”.
- Do not give “just a little bit more” until you can buy another brand.
- Treat every ByHeart Whole Nutrition infant formula product as recalled and off-limits.
Even if your baby is fine right now, you don’t keep feeding them a product that’s under a botulism investigation.
Step 2 – Label and store the product (don’t throw it in the bin yet)
You’re going to quarantine the formula, not destroy the evidence.
Do this:
- Take clear photos of:
- The front of the can or box (showing the brand and product name).
- The lot number and “use by” date (usually printed on the bottom or side).
- On the can or box, write in big letters: “RECALLED – DO NOT USE”
- Put it somewhere out of reach of children and pets.
Why keep it?
- Public-health teams sometimes ask families for lot information or leftover product.
- The company or retailer may require photos or physical return for refunds or replacements.
- Tossing it immediately helps nobody; storing it safely helps everyone.
Step 3 – Switch your baby to a different formula safely
This is where many parents panic and start trying random stuff. Don’t.
- Call your pediatrician and say clearly: “My baby has been using ByHeart Whole Nutrition infant formula, which is now recalled for an infant botulism outbreak. I need a safe alternative brand for my baby’s age and situation.”
- If you can’t reach your doctor immediately:
- Choose a well-known brand with standard cow’s-milk-based formula for your baby’s age, unless your baby already needs a special type (hypoallergenic, soy, etc.).
- Once you speak to the doctor, confirm that your temporary choice is appropriate.
- Don’t:
- Mix up homemade formula recipes from social media.
- Dilute formula more than the instructions to “make it last longer”.
- Switch to cow’s milk for very young infants without medical advice.
Remember: health authorities have been clear that ByHeart is a tiny share of the U.S. formula market, and no one is expecting another nationwide shortage because of this recall. Stores may have local gaps, but this isn’t the 2022 crisis all over again.
Step 4 – Watch your baby like a hawk for several weeks
This is where you replace vague anxiety with structured monitoring.
For at least 4 weeks after the last ByHeart feed:
- Keep a simple daily note of:
- Stools (how often, hard/soft, any new constipation).
- Feeding (amount taken, how long it takes, any fatigue or choking).
- Behaviour and movement (normal, lazier, floppy, less expressive).
- Breathing (normal vs anything that worries you).
You don’t need a fancy app; your phone’s notes app is enough. Just make it easy to tell a doctor, “This is what changed and when.”
Step 5 – Decide when to call the doctor vs go straight to ER
Call your pediatrician the same day if:
- Your baby used ByHeart, and
- You notice early signs: new constipation, weaker feeding, odd-sounding cry, mild floppiness, or less movement.
Go straight to ER / emergency if:
- Your baby is floppy like a rag doll,
- Has obvious trouble swallowing or keeping milk down,
- Shows any breathing changes,
- Is very hard to wake or unusually unresponsive.
When you arrive, be direct:
“My baby used ByHeart Whole Nutrition infant formula that is under a national recall for an infant botulism outbreak. Now they have [list symptoms]. I’m worried about infant botulism.”
That immediately puts the right diagnosis on the radar.
Step 6 – Tell your doctor the full story, not an edited version
People often downplay things because they don’t want to look anxious. That’s stupid here.
Tell them:
- How long your baby used ByHeart.
- When the last ByHeart feed was.
- When symptoms started and how they changed.
- Any other exposures (dusty home, honey, weird supplements).
You’re not “bothering” anyone. You’re giving the doctor the information they need to decide whether to call public health and push for BabyBIG.
Step 7 – After this, don’t carry guilt, carry lessons
If your baby used ByHeart, you didn’t cause this. You bought a formula that was legally on the market and marketed as safe. That’s it.
The only thing that defines you now is how you respond:
- Whether you stop the product immediately.
- Whether you watch your baby with clear eyes.
- Whether you act fast if something feels wrong.
Do those three things properly, and you’ve done your job as a parent in this situation.
How Doctors Diagnose and Treat Infant Botulism
Here’s the uncomfortable truth: there is no single magic test that instantly says “yes, this is infant botulism” in the ER. Doctors almost always move on a clinical suspicion first and let the lab results catch up later.
How doctors decide “this could be botulism”
When you walk into an ER or clinic with a baby who used ByHeart and now looks weak or isn’t feeding well, the doctor is mentally checking a few boxes:
- Age: under 12 months (especially under 6 months).
- Exposure: history of ByHeart Whole Nutrition infant formula use (now strongly associated with this outbreak) or other known sources like honey or significant dust exposure.
- Symptom pattern:
- New constipation →
- Poor feeding + weak cry →
- Floppiness / loss of head control →
- Breathing and swallowing problems.
If that triad (age + exposure + symptom pattern) fits, good clinicians don’t waste hours pretending it might just be “gas” or “teething”. They start working as if this is infant botulism and escalate appropriately.
The role of tests (stool, nerve studies, etc.)
To confirm the diagnosis, doctors and public-health labs will usually:
- Send stool samples to test for Clostridium botulinum and its toxin. This is the gold standard, but it can take days to weeks to come back from specialized state or reference labs.
- Sometimes arrange electrodiagnostic tests (nerve conduction studies/EMG) to look for patterns consistent with botulism, if available.
But crucial point:
Treatment is not delayed just because lab results are pending. If the clinical picture screams “infant botulism”, they move.
BabyBIG – the antitoxin made specifically for infants
The main specific treatment is a medicine called BabyBIG® (Botulism Immune Globulin Intravenous or BIG-IV). It’s not an everyday drug sitting on every hospital shelf; it’s ordered through specialized channels (in the U.S., historically via California’s Infant Botulism Treatment and Prevention Program).
What BabyBIG does:
- It provides ready-made antibodies that bind to circulating botulinum toxin.
- That stops new toxin from attacking the nerves, so the baby can start recovering as the damaged nerve endings slowly regenerate.
- Studies have shown it shortens hospital stay and reduces complications compared with supportive care alone.
The sooner that decision is made, the better. That’s why being upfront about ByHeart exposure and describing symptoms clearly actually matters.
Hospital care: what actually happens to the baby

Most infants with confirmed or strongly suspected botulism will be:
- Admitted to hospital, often to a PICU (pediatric intensive care unit) or high-dependency unit for close monitoring.
- Given BabyBIG as a single intravenous infusion, assuming they meet criteria.
- Watched carefully for:
- Breathing effort (sometimes needing ventilator support).
- Swallowing ability (may require feeding tube).
- Hydration and nutrition.
Recovery is not instant. Even with antitoxin, nerve endings need time to heal. That can mean:
- Weeks of gradual improvement in strength and feeding.
- Sometimes months before the baby is fully back to baseline.
The good news in this outbreak so far:
- All reported infants have been hospitalized and treated, and as of the latest updates, no deaths have been reported.
That doesn’t make it “mild” – but it does mean that early recognition and proper care work.
Will the ByHeart Recall Cause Another Baby Formula Shortage?
A lot of parents see “formula recall” and immediately flash back to the 2022 formula crisis. That’s understandable, but you need to separate emotion from facts here.
Short answer: No, this recall by itself is very unlikely to cause a nationwide shortage.
How big is ByHeart in the U.S. market?
ByHeart is a small, newer brand. According to FDA and multiple news reports, their products account for well under 1% of all U.S. infant formula sales.
So when all ByHeart Whole Nutrition formula is pulled:
- It’s a huge deal for ByHeart users.
- It’s a tiny fraction of the total formula supply in the country.
This is fundamentally different from shutting down a major plant that serves a big chunk of the national market, like what happened with the Abbott/Sturgis situation in 2022.
What FDA and states are actually saying about supply
FDA’s outbreak and recall communications have been very clear on this point:
- They do not expect this recall to trigger a national formula shortage.
- Other big manufacturers are operating normally; there’s no broad production shutdown.
Some state health departments have echoed the same thing: families affected by the recall may need to switch brands or visit a different store, but there’s no sign of a country-wide supply shock tied solely to ByHeart.
What you should actually do about formula supply
Here’s the rational approach instead of spiralling into “there’ll be no milk in the whole country” mode:
- Plan your switch early
- Don’t wait until you’re on your last scoop to look for an alternative.
- Talk to your pediatrician now about which brand is best for your baby (standard vs hypoallergenic, etc.).
- Be flexible on brand, rigid on safety
- You may not find your “first-choice” alternative on the first try, but you’ll almost certainly find some reputable brand.
- What you don’t do is:
- Order sketchy imported formula from random sellers with no proper regulation.
- Mix your own “DIY baby formula” from YouTube or TikTok recipes.
- Use official information, not rumor chains
- Check:
- FDA recall page for updates.
- State health department notices where you live (many list helplines or WIC support options).
- Ignore viral “someone said all U.S. formula is unsafe” posts. That’s noise.
- Check:
- If you’re on WIC or similar programs
- Many states adjust their WIC-approved product lists during recalls so families can swap brands without losing coverage.
- Contact your local WIC office; don’t assume you’re stuck.
Reality check
Could there be local supply hiccups—like your usual store being out of your first alternative brand because lots of ByHeart parents switched at once? Yes, that can happen in the short term.
But structurally, pulling <1% of the U.S. formula market does not equal “no formula for your baby”. It means:
- You may have to change brand.
- You may have to visit more than one store or order online.
- You absolutely do not need to keep using a recalled formula out of fear of shortage.
If you’re still tempted to ration or “stretch” a recalled can because you’re anxious about supply, that’s you letting fear override basic safety. Don’t do that. Use a safe, available alternative and leave ByHeart in the do-not-use box until this outbreak is fully resolved.
How Parents Can Reduce Botulism Risk Beyond This Outbreak
Once you’ve dealt with the ByHeart issue, the next question is:
“Okay, what can I do so this kind of thing is as unlikely as possible in the future?”
You can’t get the risk to zero, but you can remove the big, stupid risks and tighten up basic food safety around your baby.
1. No honey before 12 months – zero exceptions

This one is non-negotiable and has nothing to do with ByHeart. For decades, we’ve known:
- Honey can contain C. botulinum spores.
- Babies under 1 year don’t have mature gut defenses, so spores can germinate and produce toxin.
So:
- No honey in milk.
- No honey on pacifiers.
- No honey “for cough”.
- No herbal syrups or “natural” remedies that sneak honey into the ingredients for infants under 12 months.
If someone says “It’s just a little, it’s natural,” they’re talking nonsense. You have one job here: no honey before 1 year.
2. Handle formula like it actually matters
Botulism is rare, but bad formula hygiene can still cause other serious infections. Don’t get sloppy.
Basic rules (that most people quietly ignore):
- Wash your hands before making a bottle. Every time.
- Use clean, properly washed bottles and nipples; wash in hot soapy water, rinse well, and let them air dry.
- Follow the mixing instructions exactly:
- Correct powder-to-water ratio (no “stretching” to save money).
- Use safe water according to local guidance.
- Don’t leave prepared formula sitting out:
- Use within 2 hours of preparation, or
- Within 1 hour of starting to feed.
- Discard leftovers from a feed; don’t put half-finished bottles back in the fridge “for later”.
None of this is glamorous, but this is real-world risk reduction.
3. Be picky about “alternative” foods and trendy stuff
Infants don’t need fancy powders and wellness junk. In fact, it adds risk for zero benefit.
Avoid for babies under 1 year:
- Unregulated “superfood” powders, random herbal mixes, or imported porridges with no proper safety oversight.
- Home-fermented products (kombucha, weird “probiotic” brews) and “raw” products meant for adults.
Stick to:
- Breast milk where possible.
- Commercial infant formula from reputable brands with active regulatory oversight.
- Age-appropriate solids recommended by your pediatrician.
You are not depriving your baby by skipping Instagram health hacks. You’re protecting them.
4. Keep the environment reasonably clean without going insane
You don’t have to turn your house into an operating theatre, but you also don’t ignore obvious contamination.
Common-sense steps:
- If you live in a very dusty, construction-heavy, or agricultural area, try not to let your baby play directly on obviously dusty floors or soil where they constantly put contaminated hands in their mouth.
- Regularly clean surfaces where your baby crawls or plays.
- Don’t store baby bottles or open formula cans in damp, dirty corners of the kitchen.
This doesn’t “guarantee” anything, but it keeps the exposure level sensible.
5. Learn the patterns of serious illness instead of googling every sneeze
You can’t memorize every disease, but you can remember a few dangerous patterns, including the one for botulism:
- New constipation + poor feeding + floppy baby + weak cry + breathing issues
→ ER, now.
The same principle applies to other emergencies (severe dehydration, meningitis signs, etc.). Developing this mental library is more useful than binge-watching fear-based content.
For Health Professionals and Public Health Students
If your site is read by clinicians, residents, or MPH students, this is where you quietly show you know what you’re talking about.
Why this outbreak matters for food safety and surveillance
Three big public-health points make this incident more than just “one more recall”:
- Unusual vehicle
- Infant botulism is typically sporadic and linked to environmental dust or honey, not commercial formula.
- A cluster tied to infant formula forces regulators to revisit assumptions about “low-risk” products for vulnerable groups.
- Signal detection in a rare disease
- With a very low baseline incidence, even a modest increase triggers suspicion.
- Here, combining data from multiple states + California’s Infant Botulism Treatment and Prevention Program allowed early pattern recognition.
- Balancing incomplete lab data with strong epidemiology
- Regulators moved on a strong epidemiologic association and preliminary product testing, even before finding toxin in unopened cans.
- This is textbook precautionary action in high-severity, low-incidence events.
If you’re in public health, this is a live example of risk management under uncertainty, not textbook perfection.
Clinical reminders for doctors and trainees
For clinicians, especially in pediatrics, EM, and primary care, this outbreak is a blunt reminder:
- Think infant botulism in any infant under 1 year with:
- New constipation
- Descending weakness
- Poor feeding and weak cry
- Cranial nerve involvement (poor suck, facial weakness)
- ± known exposure to ByHeart formula or honey.
- Don’t sit on it.
- Call your local/state health department early.
- In the U.S., get in touch with the Infant Botulism Treatment and Prevention Program for guidance and BabyBIG procurement.
- Avoid unnecessary interventions
- No aminoglycosides or other agents that worsen neuromuscular transmission.
- Avoid magnesium-containing laxatives and certain neuromuscular blocking agents unless absolutely necessary and under specialist guidance.
- Document formula brand and lot
- For any floppy infant or suspected botulism case, explicitly record formula type, feeding history, and lot details if formula is used.
- That information is not just clinical; it’s surveillance data.
Public-health practice takeaways
For MPH / public-health audiences, you can spell out a few system-level lessons:
- Surveillance: rare but severe syndromes (like infant botulism) need centralized expertise and real-time aggregation of cases across states.
- Communication: mixed messaging from industry vs regulators confuses the public; clear, firm public-health messaging matters more than corporate damage control.
- Equity: families on WIC or low income are more exposed to stress from recalls; planning for rapid brand substitution and support is part of outbreak management, not an afterthought.
You don’t need to write a whole thesis. A few sharp paragraphs like this send a clear signal: your article isn’t just recycled health content; it’s grounded in how real systems and real clinicians actually work.
Key Takeaways for Busy Parents
If you skimmed everything else, read this part slowly and carefully:
- There is an ongoing infant botulism outbreak in the U.S. linked to ByHeart Whole Nutrition infant formula.
- As of mid-November 2025, 23 infants in 13 states with suspected/confirmed infant botulism have been reported; all hospitalized, no deaths so far.
- All ByHeart Whole Nutrition infant formula products (all lots, cans, and Anywhere Packs™) are under nationwide recall. Treat every unexpired ByHeart product as do-not-use.
- Infant botulism is rare but serious. Key warning signs in a baby who used ByHeart recently:
- new or persistent constipation,
- poor feeding or tiring at the bottle,
- weak / different cry,
- floppy head or limbs,
- changes in breathing or difficulty swallowing.
- If you see any floppiness, breathing trouble, or swallowing problems, you go straight to ER, and clearly say your baby used ByHeart formula under recall and you’re worried about infant botulism.
- This recall alone is not expected to cause a nationwide formula shortage because ByHeart is a tiny share of the U.S. market. You do not need to keep using a recalled formula out of fear of running out of milk.
- Your job now is simple:
- Stop the recalled product.
- Quarantine it (label “DO NOT USE”, keep photos and lot info).
- Switch to a safe alternative brand in consultation with your pediatrician.
- Watch your baby closely for several weeks and seek care early if anything feels wrong.
You can’t undo past feeds. You can absolutely control what you do from today onwards.
FAQs About the ByHeart Infant Botulism Outbreak
You can use these as your FAQ block and wrap them in FAQ schema for SEO.
1. Is ByHeart formula safe to use right now?
No. All ByHeart Whole Nutrition infant formula (cans and Anywhere Pack™ sticks, all lots, all unexpired product) is under nationwide recall because of a multistate infant botulism outbreak. Treat every unexpired ByHeart formula product as unsafe for infant feeding until CDC/FDA say otherwise.
2. Which ByHeart products are included in the 2025 recall?
The recall covers:
- ByHeart Whole Nutrition Infant Formula – 24 oz powdered cans
- ByHeart Whole Nutrition Infant Formula Anywhere Pack™ single-serve sticks
All unexpired lots and all sizes are affected. If the label says ByHeart Whole Nutrition Infant Formula, assume it’s recalled.
3. My baby finished a can of ByHeart weeks ago and seems fine. Do I still need to worry?
You don’t need panic, but you are not automatically “safe” either. Infant botulism symptoms can appear days to several weeks after exposure.
If your baby used ByHeart in the last few weeks:
- Do not give them any more.
- Watch closely for: constipation, poor feeding, weak cry, floppiness, or breathing changes.
- If any of these appear, call your pediatrician urgently or go to the ER.
If your baby remains completely well over several weeks, the risk becomes very low.
4. How long after using ByHeart formula can infant botulism symptoms appear?
Symptoms typically show up within days to several weeks after spores are swallowed. For practical purposes, you should monitor your baby for at least 4 weeks after their last ByHeart feed for any of the warning signs listed in this article.
5. What is usually the first sign of infant botulism?
In many infants, the first noticeable sign is new or worsening constipation, followed by:
- poor feeding,
- weaker or “different” cry,
- and gradually increasing floppiness.
Any combination of new constipation + feeding issues + weakness in a baby who used ByHeart recently is a reason to seek medical care quickly.
6. Should I switch my baby to a different formula brand immediately?
Yes. If you’re using ByHeart, you should stop it today and switch brands.
- Call your pediatrician to choose the right alternative (standard cow’s-milk formula vs hypoallergenic, etc.).
- Do not use homemade formula recipes or random imported products from unverified sellers.
You can switch first to a reputable mainstream brand and then fine-tune with your pediatrician if needed.
7. Can infant botulism be cured? What is the prognosis?
Most babies survive and eventually recover, especially if treated early.
- The main specific treatment is BabyBIG® (botulism immune globulin), which neutralizes circulating toxin.
- Many infants need hospital and sometimes ICU care for feeding and breathing support.
- Recovery can take weeks to months, but long-term outcomes are generally good when managed properly and promptly.
8. Is this outbreak causing a new baby formula shortage in the U.S.?
No. Health authorities do not expect this recall alone to trigger a nationwide shortage.
- ByHeart accounts for well under 1% of U.S. infant formula sales.
- Major manufacturers and other brands are still operating normally.
You may have to switch brands and possibly check more than one store, but this is not a repeat of the 2022 large-scale shortage.
9. Can breastfeeding babies also get infant botulism?
Yes, but it’s rare. Infant botulism has been reported in breastfed babies as well, usually from environmental exposure (dust/soil) or honey given before 12 months.
In this outbreak, the main concern is formula-exposed infants who used ByHeart. If your baby is exclusively breastfed and has never had ByHeart or honey, their risk from this specific recall is extremely low.
10. Where can I get official updates about the ByHeart recall and outbreak?
For current, verified information, rely on:
- CDC – Infant botulism outbreak page
- FDA – ByHeart recall and outbreak investigation page
- Your state health department website
These sources are updated as new cases or test results appear and are far more reliable than social media rumors.


