Chagas disease in the U.S. is gaining attention as researchers document widespread kissing bugs, infected animals, and sporadic locally acquired cases. This 2025 guide brings everything into one place: a U.S. states map, how infection really happens, symptoms to watch for, who should get tested, current treatment options, and practical prevention for homes and kennels. It’s written for readers who want clear, trustworthy answers based on CDC and peer-reviewed sources.
1) Is Chagas now considered endemic in the U.S.?
Short answer: the scientific case for calling Chagas endemic in the United States is strong and current. A 2025 perspective in CDC’s journal Emerging Infectious Diseases reviews decades of evidence—widespread kissing bug (triatomine) populations, infected wildlife and dogs acting as reservoirs, and locally acquired (autochthonous) human cases in multiple states—and concludes the U.S. meets criteria for at least hypoendemic status. Reframing it this way matters because “non-endemic” labeling has historically depressed surveillance, clinical awareness, and testing. CDC Travelers’ Health
What this means for readers: Chagas isn’t only an imported issue tied to travel. Triatomine bugs are established across the southern U.S., they occasionally enter homes and kennels, and sporadic local transmission has been documented—Texas has reported cases every year from 2013–2023. At the same time, CDC materials still describe U.S. autochthonous cases as “small in number,” which underscores two realities: the risk is not uniform, yet the disease is under-recognized and easy to miss without targeted testing. CDC Travelers’ Health+1
2) States map—where kissing bugs and cases are reported
If you’re looking for a reliable U.S. states map, the Texas A&M Kissing Bug team maintains the best public, continuously updated view. Their interactive map shows where kissing bugs have been found or submitted by the public (29+ states), with an important caveat: submissions ≠ risk. Absence of dots doesn’t prove absence of bugs; it often reflects under-sampling. Use this map for presence and seasonality context, not precise risk prediction. kissingbug.tamu.edu+1
For a public-health view, cite the CDC journal figure that layers three things on one U.S. map: 1) states with triatomines, 2) states with infected animals (wild/captive/companion), and 3) states with documented locally acquired human cases. That visual helps readers grasp why scientists now argue for endemic classification—vectors and reservoirs are widespread, and human cases, while uncommon, occur repeatedly (with Texas the most notable). Pair this with CDC’s overview map of triatomine species in the southern U.S. for additional authority.
U.S. Chagas (Kissing Bug) Map — Presence, Animal Positives & Locally Acquired Cases
Presence ≠ uniform risk. Use this map for awareness; see sources below for details.
State-by-state overview
| State | Presence | Animals | Human cases |
|---|
Sources: CDC EID 2025 (U.S. endemicity) · CDC EID Appendix (per-state table) · Texas A&M interactive map
3) How infection happens (and common misconceptions)

The short version: Chagas is caused by the parasite Trypanosoma cruzi. In the U.S., the main route is contact with the feces of an infected kissing bug (triatomine)—not the bite itself. After feeding, the bug may defecate near the bite. If that material is rubbed into the tiny puncture, into the eye, or onto broken skin, infection can occur.
All known routes (U.S.–relevant):
- Vector-borne: contaminated bug feces introduced into the bite site, eye (Romaña sign risk), or a skin break.
- Congenital: an infected mother can pass T. cruzi to the baby during pregnancy.
- Blood transfusion & organ transplant: rare due to screening, but still possible when safeguards miss a case.
- Laboratory & accidental exposures: uncommon, occupational.
- Food-borne: documented in Latin America via contaminated juices/foods; very rare in the U.S.
What does not spread Chagas:
- Casual contact, hugging, sharing utensils, or airborne exposure.
- Contact with healthy-appearing pets. Dogs can be infected reservoirs, but they don’t directly “give” Chagas to people by licking or cuddling. The risk is environmental—kennels, porches, woodpiles—where bugs and their feces may be present.
Household risk patterns to know (SEO quick hits):
- “Where do kissing bugs live?” → cracks/crevices of walls, attics, crawl spaces, dog kennels, chicken coops, outdoor lighting areas.
- “Do kissing bugs live in beds?” → they can enter homes and hide near sleeping areas, but U.S. infestations are typically peridomestic (around the home) rather than heavy indoor colonization.
- “How to reduce risk” → seal entry points, manage outdoor lighting, remove brush/wood piles, elevate kennels, and follow safe collection steps if you find a suspect bug.
4) Symptoms—acute vs. chronic (when to test)

Incubation & phases:
- Acute phase (weeks–months after exposure): Many people have no symptoms or only mild, flu-like illness.
- Chronic phase (lifelong without treatment): Most remain indeterminate (no organ disease), but ~20–30% can develop heart or gastrointestinal complications years later.
Acute symptoms to watch for (often nonspecific):
- Fever, fatigue, body aches, headache
- Swollen lymph nodes; mild liver/spleen enlargement
- Chagoma (swollen, sometimes painful skin lesion at entry site)
- Romaña sign (painless swelling of one eye if contamination occurred there)
Chronic disease (develops in a subset over years):
- Cardiac: conduction abnormalities (e.g., right bundle branch block), arrhythmias, cardiomyopathy, heart failure, stroke/embolism
- Gastrointestinal (less common in U.S.): difficulty swallowing, chronic constipation, megacolon/megaesophagus
When to consider testing for Chagas (practical checklist)
You should talk to a clinician about testing if any of the following apply:
- Exposure + symptoms: You live(d) or spend a lot of time in U.S. regions with kissing bug presence and had a compatible illness or Romaña sign after possible exposure.
- Birth or long residence in Latin America where Chagas is endemic—especially if you’ve never been tested.
- Pregnancy with personal risk factors (born/long-term in endemic regions) or if your mother is from an endemic area and may not have been screened.
- Abnormal heart findings without a clear cause (unexplained cardiomyopathy, new conduction blocks/arrhythmias) and any epidemiologic risk.
- Blood donor notification that your screening was reactive for T. cruzi antibodies.
- Organ transplant context (donor or recipient) with relevant risk factors.
What to expect if you test:
- Acute infection: clinicians may use PCR plus serology.
- Chronic infection: diagnosis usually requires two different antibody tests; a reference lab may confirm discordant results.
- Why early matters: Treatment is most effective as early as possible, especially in acute and early chronic infection and in congenitally infected infants (the next section of this guide covers treatment options and access).
Helpful tip for readers: Keep a quick “exposure diary”—where you live(d), any known kissing bug encounters, travel history, blood donation flags, and heart test results. Bringing this to your appointment speeds clinical decision-making and reduces the risk of missed diagnosis.
5) Testing & diagnosis in the U.S. (what actually happens)
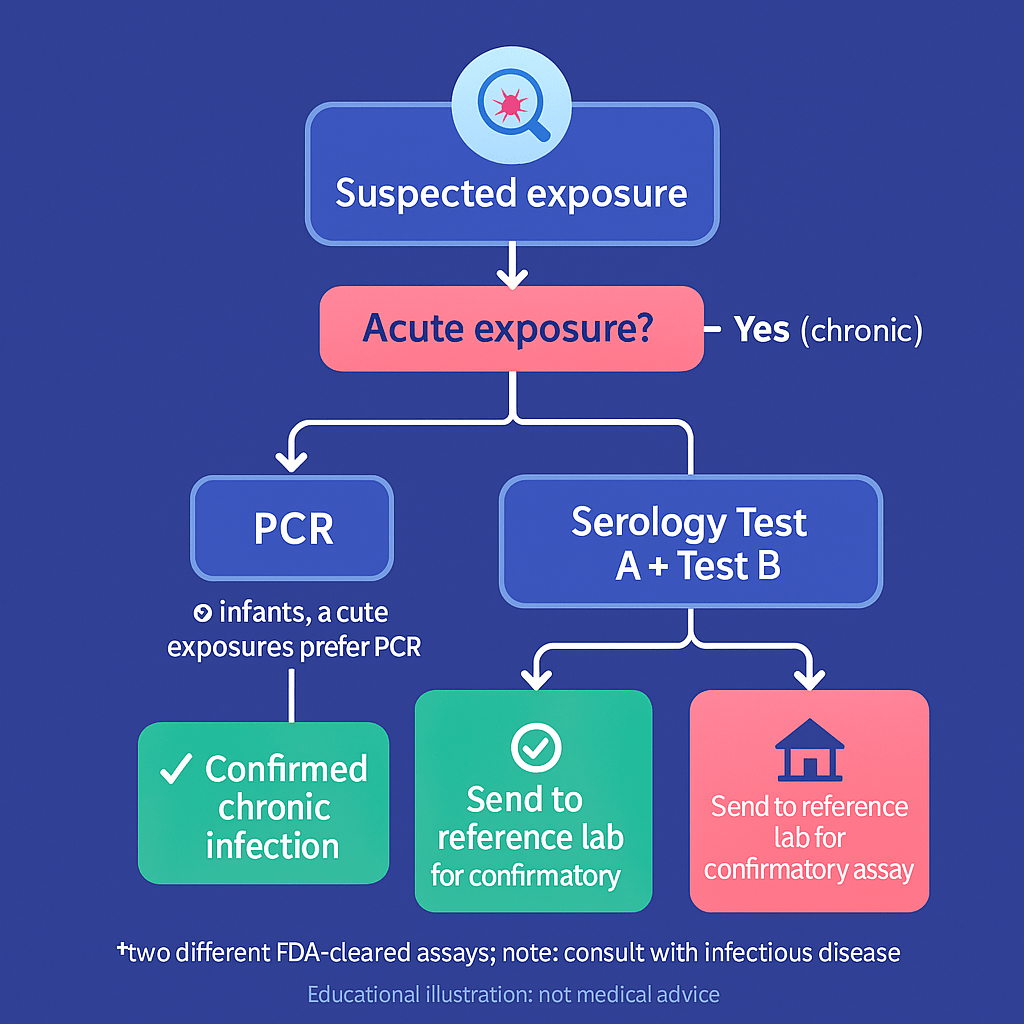
Bottom line: most people are diagnosed with blood tests (serology); in early/acute infection doctors may also use PCR to look for parasite DNA.
How clinicians confirm it
- Acute or very recent exposure: PCR (and sometimes parasite visualization) can detect infection early, when antibody tests may still be negative.
- Chronic infection (the most common scenario in the U.S.): you need two different antibody tests (e.g., ELISA + IFA) performed on the same sample or in sequence. If results disagree, a third assay from a reference lab is used to resolve.
- Newborns/infants of at-risk mothers: test early with PCR, then repeat serology around 9–10 months (to avoid detecting the mother’s antibodies).
- Blood donor notification: U.S. blood centers do one-time Chagas screening. If you’re told your donation was reactive, the blood bank (or your clinician) will arrange confirmatory testing and next steps.
Where to get tested
- Start with your primary care physician or an infectious diseases clinic; they can send samples to commercial or reference laboratories that run validated Chagas assays.
- Your state health department can advise on reference testing pathways and follow-up.
Pro tips for readers (SEO-friendly FAQs)
- Can I rely on a single “Chagas test”? → No. For chronic disease, diagnosis requires two distinct antibody tests.
- How long do results take? → Routine serology returns in days; confirmatory testing can take longer.
- What if I found a kissing bug? → Don’t crush it. Safely collect in a sealed container; follow your state’s guidance or university extension program for ID/submission.
6) Treatment — benznidazole & nifurtimox (what patients should know)
Goal of therapy: kill or suppress T. cruzi to prevent disease progression and reduce transmission (especially in acute, congenital, and early chronic infection). Decisions for long-standing chronic cases are individualized.
The two antiparasitic options
- Benznidazole — long used worldwide; FDA-approved in the U.S. (pediatric label) and commonly prescribed for adults based on clinical judgment.
- Nifurtimox (Lampit) — FDA-approved (pediatric label); also used for older adolescents/adults at a clinician’s discretion.
Who benefits most (typical priorities)
- Acute or reactivated infection (including after transplant)
- Congenitally infected infants (highest cure rates when treated early)
- Recent or early chronic infection (often up to middle age, case-by-case)
What treatment is like
- Given by mouth for several weeks (exact regimen chosen by your clinician).
- You’ll have baseline labs (e.g., liver function) and periodic checks during therapy.
- Side effects are common but usually manageable:
- Benznidazole: rash, itching, headache, nausea; occasionally neuropathy or liver enzyme bumps.
- Nifurtimox: appetite loss, nausea/abdominal discomfort, trouble sleeping, irritability; rarely neuropathy.
Tell your doctor promptly if you develop a rash, numbness/tingling, severe GI symptoms, or yellowing of eyes/skin.
After treatment
- You won’t get a quick “negative” blood test—antibodies can stay positive for years. Doctors follow symptoms, ECG/echo (heart), and sometimes serial PCR or evolving serology.
- Keep your exposure diary (travel, housing, bug encounters, prior test results); it helps guide long-term follow-up.
Access & practical notes
- U.S. pharmacies can dispense both medicines; some manufacturers and nonprofits offer patient assistance programs.
- Therapy doesn’t replace prevention—seal entry points, manage outdoor lighting, kennel hygiene, and follow local guidance on bug submissions (see the map section).
7) Pets & household risk (dogs as “sentinels,” not direct spread)
Key idea: Dogs (and some wildlife) can get T. cruzi and signal that kissing bugs are active around a home—but pets don’t “give” Chagas to people by casual contact. The risk is environmental: places where bugs hide, feed, and defecate (kennels, porches, coops, woodpiles).
What raises risk around homes
- Peridomestic shelters: outdoor dog kennels, porches, sheds, chicken coops, stacked lumber/brush.
- Night lighting: bright, unshielded bulbs near doors/windows can attract insects, including triatomines.
- Gaps & cracks: unscreened vents, loose window/door screens, unsealed siding/rooflines.
Practical steps for pet owners
- Kennel hygiene & layout: elevate kennels off soil; seal cracks; add fine-mesh screening; place away from exterior walls and lights; keep bedding clean and dry.
- Bring pets in at night where feasible; feed indoors; store pet food in sealed containers.
- Yard housekeeping: remove brush/woodpiles; trim vegetation touching structures; relocate coops/sheds away from the house.
- If you find a suspect bug on/near a pet: don’t crush it; use gloves/tweezers to place it in a sealable container (with alcohol if advised locally). Wash hands and surfaces.
- Vet conversation: if you live in a known kissing-bug area or your dog had a compatible illness, ask your veterinarian about risk, testing options, and follow-up. (There’s no vaccine for dogs; management is case-by-case.)
8) Prevention at home — a quick, practical checklist

Think layered protection: seal the house, manage the yard, handle bugs safely. Use these bullets as a scannable checklist for readers (and a featured snippet target).
A. Seal & screen
- Weather-strip exterior doors; install door sweeps.
- Repair/replace torn window screens; add fine mesh to attic/crawl-space vents.
- Caulk cracks around siding, utility lines, eaves, and foundation gaps.
B. Smart lighting
- Swap bright white bulbs by entries for warm-spectrum or yellow “bug” lights.
- Use motion-activated, downward-shielded fixtures; keep lights away from doors/windows when possible.
C. Yard & structures
- Move woodpiles/brush away from buildings; keep lawns trimmed and clutter down.
- Elevate and screen kennels/sheds/coops; clean weekly; avoid placing them against house walls.
- Fix loose roofing, siding, and fascia boards where insects can shelter.
D. Sleeping areas (home & travel)
- Pull beds a few inches off walls; tuck bedding; consider bed nets when camping or in higher-risk settings.
- In warmer months, close unscreened windows at night.
E. Safe bug handling
- If you suspect a kissing bug: don’t crush with bare hands. Capture into a jar or zipper bag (use gloves/tweezers), note the location/date, and follow your state/university extension’s submission guidance.
- Clean the area; avoid touching possible bug feces; wash hands.
F. When to call a pro
- For recurring finds or suspected harborage, contact a licensed pest management professional familiar with triatomines. Ask for an integrated pest management approach and strictly follow all label instructions for any insecticides.
9) Kissing bug ID (with look-alikes)
What a true triatomine looks like (quick checklist):
- Head: long, narrow “cone-nose”; beak (rostrum) points straight forward.
- Body edges: alternating light/dark stripes along the outer abdomen (“connexivum”).
- Size & wings: ~1–3 cm; adults have flat wings crossing on the back (nymphs are wingless).
- Antennae & legs: long, slender; no “leafy” expansions.
If in doubt, compare with CDC’s species/ID pages, which show close-ups of U.S. triatomines. (CDC)
Common look-alikes (not Chagas vectors):
- Stink bugs / squash bugs / western conifer seed bug: shield-shaped or with a zig-zag line; many have leaf-like hind legs.
- Wheel bug (an assassin bug): large gear-shaped crest on the back—not a kissing bug.
CDC’s “Bugs commonly confused with triatomine bugs” gallery is perfect for side-by-side checks. (CDC)
If you think you found one: don’t crush it. Use gloves/tweezers; place it in a sealed container, note date/location (indoors/outdoors), and follow testing/ID directions from the Texas A&M “Found a Bug?” page or your state extension program. (kissingbug.tamu.edu)
10) Special situations
A) Pregnancy & congenital transmission (screening advice)
- Risk: An infected mother can pass T. cruzi to the baby during pregnancy; many infants are asymptomatic, so testing is essential. (CDC)
- Testing plan: PCR for the newborn in the first weeks/months, with confirmatory serology at ~9–10 months (to avoid maternal antibody interference). Evaluate and test other children if indicated. (CDC)
- Treatment timing: Antiparasitic therapy for infants is highly effective; treatment of mothers is typically deferred until after delivery and individualized by clinicians. Breastfeeding is generally safe; if nipples are cracked/bleeding, pump and discard until healed. (CDC)
B) Blood donation — can I donate if I’ve had Chagas?
- Screening: U.S. blood centers perform one-time donor screening for T. cruzi; reactive results trigger confirmatory testing and counseling. (AABB)
- Deferral basics: Historically, repeat-reactive donations are quarantined and donors deferred; policies follow FDA/AABB standards—ask your blood center for your specific status and re-entry options. (CDC)
- If you’re notified: share the letter with your clinician and pursue confirmatory testing; this page’s Testing & Diagnosis section explains the two-assay algorithm. (CDC Travelers’ Health)
C) Travelers & immigrants — what to discuss with a clinician
- Who should consider testing: anyone born in or with long residence in Latin-American endemic regions, or travelers with rural/night-time vector exposure—especially if there are cardiac findings (new conduction blocks/arrhythmias, idiopathic cardiomyopathy). (CDC)
- What to ask for: the two-test serology approach for chronic infection; PCR only helps in acute/very recent exposure or for infants. Bring travel/residence history, bug encounters, and any blood-bank notifications to the visit. (CDC Travelers’ Health)
11) Public-health actions & One-Health context (what’s being done—and what you can do)
Why One-Health? T. cruzi cycles among insects, wildlife, dogs, and people. Managing risk means coordinating human health, veterinary health, and the environment.
Federal & state actions (what agencies focus on)
- Surveillance & mapping: tracking triatomine species, animal positives, and sporadic locally acquired human cases; refining state guidance based on seasonality and habitat.
- Blood safety: U.S. blood banks perform one-time Chagas screening for donors, with confirmatory testing pathways and donor counseling.
- Congenital screening pathways: encouraging testing in at-risk pregnancy contexts and clear follow-up of infants born to infected mothers.
- Lab & clinician support: reference-lab confirmation, updated testing algorithms, and CME resources so primary care, cardiology, OB/GYN, and ID teams recognize risk patterns.
- Public education: “what to do if you find a kissing bug,” safe handling, and home-proofing steps.
Community & built-environment measures
- Integrated Pest Management (IPM): sealing exterior gaps; repairing screens/vents; managing outdoor lighting; keeping kennels/coops elevated and screened; brush/woodpile removal; targeted insecticide use per label by licensed professionals.
- Housing quality: attention to rural and peri-urban homes where triatomines can shelter in wall/roof gaps or outbuildings.
Health-system playbook (quick wins)
- Add a Chagas risk prompt in EMR problem-lists for patients with: long residence in endemic regions, unexplained cardiomyopathy/arrhythmias, or reactive blood donor notifications.
- Create a testing reflex order set (two different serologic assays; PCR for acute/infant cases).
- Build a care pathway for positive results: treatment access, counseling, and family testing when indicated.
Data gaps & research priorities
- Better point-of-care tests, especially for early infection.
- Standardized case definitions for U.S. autochthonous transmission.
- Dog sentinel studies and wildlife ecology to target interventions.
- Climate/land-use modeling to anticipate vector range and seasonality.
How readers can help (practical)
- Submit suspected bugs to your state or university extension program; include date, location, and photos.
- If you get a blood-bank notification, follow through on confirmatory testing and share results with your doctor.
- Share this guide with neighbors who keep outdoor dogs/coops; simple yard changes lower everyone’s risk.
12) FAQs
No. Outside of congenital transmission, blood transfusion, or organ transplant, Chagas doesn’t spread person-to-person or through casual contact.
Confirmed triatomines occur across many southern and some mid-latitude states. See the interactive map above and your state’s guidance for the latest.
Don’t crush it. Using gloves/tweezers, place it in a sealable container (or bag), note the date/location, and follow your state/university submission instructions. Wash hands and the area.
Dogs can be infected and act as sentinels of environmental risk, but they don’t transmit Chagas by licking/cuddling. The risk is from kissing bugs and their feces in shared spaces (kennels, porches).
No vaccine is available. Prevention relies on home sealing, lighting management, kennel hygiene, and safe bug handling.
Early treatment (acute, congenital, early chronic) offers the best chance to clear infection and prevent complications. In long-standing chronic infection, treatment decisions are individualized—talk to an infectious-diseases clinician.
Yes—one-time donor screening is standard. If your donation is reactive, you’ll be contacted for confirmatory testing and counseling.
If you have epidemiologic risk (e.g., long residence/birth in an endemic region or significant exposure history), discuss testing with your obstetrician; infants born to infected mothers need follow-up.
References (authoritative, up-to-date)
- CDC – Emerging Infectious Diseases (Sept 2025): “Chagas Disease, an Endemic Disease in the United States.” Overview of U.S. vectors, animal reservoirs, and autochthonous human cases; argues for hypoendemic classification; includes per-state figure. CDC Travelers’ Health
- Figure 1 (updated Aug 26, 2025): U.S. states with triatomines, animal positives, and locally acquired human cases (use this to justify the map layers). CDC Travelers’ Health
- CDC – Chagas Disease hub (About, How it spreads, Prevention, Treatment, HCP Clinical Care). Use these for all patient-facing definitions, routes, testing algorithms, and drug access. CDC+4CDC+4CDC+4
- CDC – DPDx (American trypanosomiasis): lab diagnosis and parasite background. CDC
- Texas A&M Kissing Bug Program:
- Program home + interactive map (citizen submissions/presence by state) — cite when describing “states map” and seasonality. kissingbug.tamu.edu+1
- “Found a Bug?” handling/ID guidance — cite anywhere you advise safe capture/ID. kissingbug.tamu.edu
- CDC – “Bugs Commonly Confused with Triatomine Bugs” + HCP species pages — cite in the “Kissing bug ID (look-alikes)” section. CDC+1
- Blood safety (for “Special situations: blood donation”):
- AABB: Chagas disease donor screening (summary of current U.S. practice: one-time licensed antibody test for allogeneic donors). AABB
- FDA Guidance (Chagas, 2010—still referenced) confirming the one-time testing framework; useful as historical/regulatory context. U.S. Food and Drug Administration+1

